Supplemental Information
Att. L Supplemental Information..docx
Understanding Health System Approaches to Chronic Pain Management
Supplemental Information
OMB: 0920-1374
Attachment L – Supplemental information.
Exhibit 1 presents examples of guidelines at different levels that may be relevant, including the CDC Guideline for Prescribing Opioids for Chronic Pain.
Exhibit 1. Examples of Policies and Guidelines
Policy/Guideline Level |
Description |
Federal |
|
State |
|
Local: municipal, county |
|
Health System/ Organization |
|
Exhibit 2 presents a logic model of the implementation of guidelines and policies
Exhibit 2. Logic Model of Health Systems’ Implementation of Opioid Policies or Guidelines
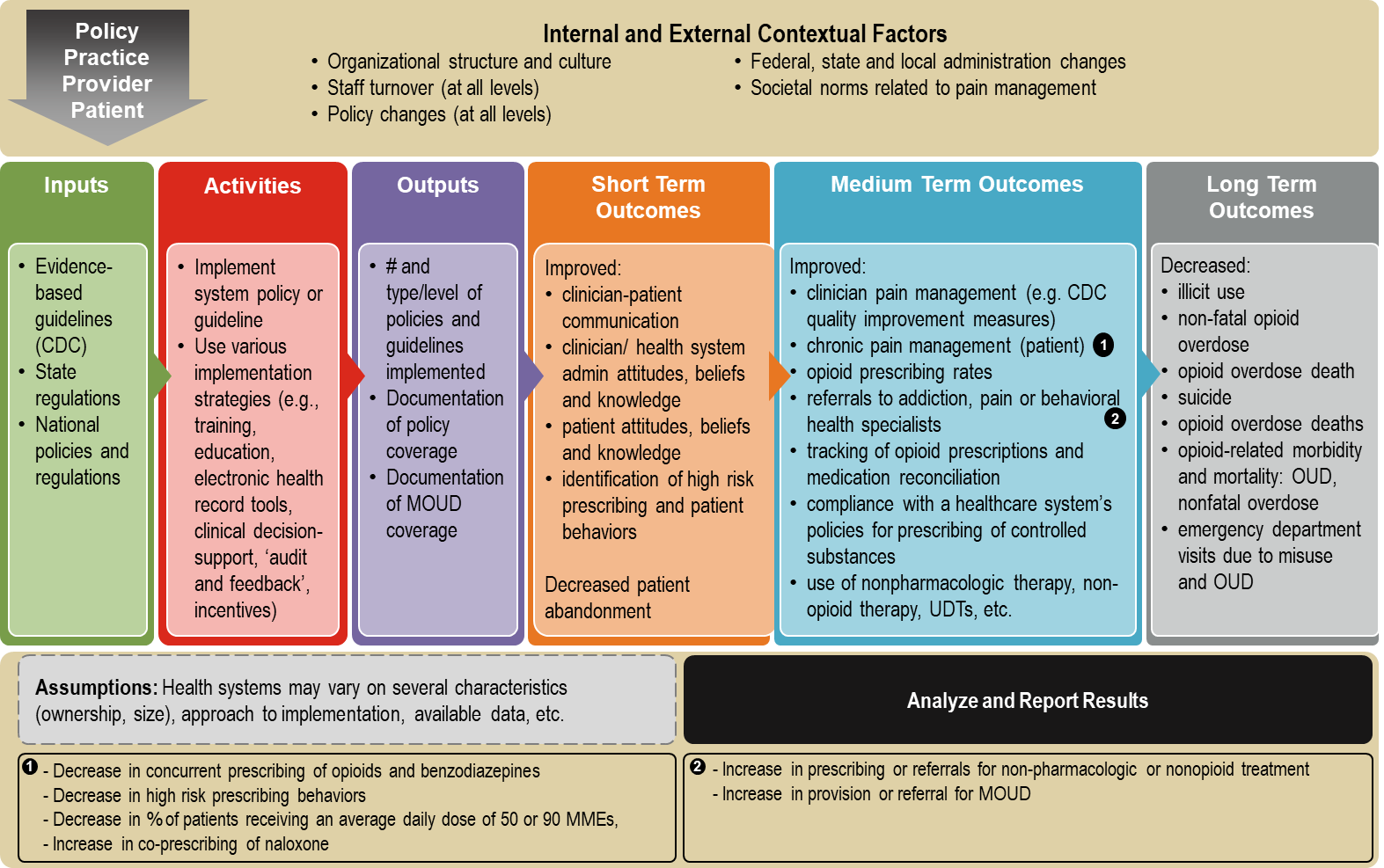
Exhibit 3 outlines the formative research questions, their data source(s), and the intended use of the data obtained from each question.
Exhibit 3. Summary of Formative Research Questions, by Data Source
Formative Research Questions |
Patient-level data |
Systems QI Measures |
Clinician Survey |
Patient Survey |
Leader Interviews |
Case Studies |
Intended Use: To identify which chronic pain management and opioid prescribing policies and guidelines were most commonly implemented and to understand health systems’ approaches and lessons learned |
|
|
|
|
X |
|
Intended Use: To determine if the implementation of these policies or guidelines resulted in changes in patient outcomes, and if these outcomes varied between patient demographic characteristics |
X |
|
|
|
|
|
Intended Use: To identify which and to what extent these policies or guidelines have directly resulted in practice change and in turn patient outcomes |
|
X |
|
|
|
|
Intended Use: To identify policies or guidelines that improved or impeded clinician-patient communication |
|
|
X |
X |
|
X |
Intended Use: To identify areas for enhanced translation and communication about guidelines and policies |
|
|
X |
X |
X |
|
Intended Use: To identify factors that influence effective uptake and implementation of policies or guidelines |
|
|
X |
X |
X |
|
Intended Use: To better understand health system-level drivers of success or failure in guideline/policy implementation efforts |
|
|
X |
X |
X |
X |
Intended Use: To identify ways in which implemented guidelines/policies may address known population-level racial/ethnic disparities in chronic pain management, opioid prescribing, and access to MOUD |
|
|
|
|
X |
X |
Description of previous work conducted.
To design this formative research study, we previously conducted and completed the exploratory work “Feasibility Assessment of Health Systems (Surveys)” to determine the range of policies and guidelines being implemented by health systems. We also completed an “evaluability assessment” by means of interviews with leaders of nine health systems, both described below.
Feasibility Assessment of Health Systems: “Feedback on the Use of the CDC Guideline for Prescribing Opioids for Chronic Pain”
In early 2020, CDC requested OMB approval for a Feasibility Assessment of Health Systems (“Feedback on the Use of the CDC Guideline for Prescribing Opioids for Chronic Pain”) through the “Generic Clearance for the Collection of Routine Customer Feedback” (OMB Control Number: 0920-1050). This brief eligibility assessment, consisting of surveys, was sent to approximately 250 health systems to understand the landscape of health systems and the types of guidelines or policies implemented, and what strategies were used to do so.
The feasibility assessment included questions in the following domains:
Health system characteristics and structure (e.g., ownership, geography, size, integration)
Levels/types of chronic pain management policies health systems have or are planning to implement (e.g., opioid prescribing, MOUD)
The stage of implementation of each policy/guideline or when they were implemented
The primary strategies for implementing polices/guidelines (e.g., policy only, training/ education, incentives, EHR and/or electronic clinical decision support (CDS) tools, use of quality improvement (QI) measures, ‘audit and feedback’, etc.)
Data systems and availability (e.g., access to full EHR reports, savvy IT analytics team, access to claims and type of claims, structured data for capturing care processes)
Of 250 health systems contacted, 46 responded and were considered for the following preliminary phase, the evaluability assessment.
Evaluability Assessment of Health Systems (Interviews)
Among the 46 health systems who completed the feasibility assessment surveys, nine were selected for a more in-depth “evaluability assessment” based on a number of factors identified in the initial feasibility (survey) assessment, as well as other expert knowledge of potential systems.
Specific criteria used to select the nine health systems for interviews included:
Years of guideline implementation: at least three years to ensure sufficient time for full implementation (i.e., implementation occurred 2017 or earlier)
Broad range of specific guideline recommendations implemented
Geographic representation across the United States
Mix of urban and rural population served, including racial, ethnic and socioeconomic variation
Presence of primary care clinicians who prescribe opioids for chronic pain management (as opposed to prescribing solely by pain management specialists)
Availability of structured data on physician practices and patient outcomes related to specific guidelines, from before and after implementation
Membership of health systems in AMGA that have expressed interest in participating and/or participated in previous research projects
We excluded systems that implemented guidelines more recently (defined as initiation of guidelines in 2018 or later) and therefore had insufficient time to observe changes over time, those with no primary care clinicians who prescribed opioids, and those without structured data.
This evaluability assessment of the selected nine systems consisted of interviews involving a more in-depth discussion of each system’s chronic pain management, opioid prescribing, and MOUD policy and guideline implementation as well as availability of data. Interviews were conducted by telephone or on a virtual meeting platform with a key informant from each system (e.g., lead for quality and/or safety in ambulatory settings, chief medical officer, or other leaders in the systems) who were knowledgeable about the specific policies and guidelines implemented, including implementation strategies (e.g., communications and trainings), timeline for implementation, barriers or facilitators to implementation, and availability of data regarding implementation of MOUD, special populations (e.g., race, ethnicity, and income), and patient outcomes. The interviews were recorded following informed consent from participants. Each interview was summarized in a Word document based on information from the notes and, when needed, the recording. All data was stored on Abt Associates’s secure server and will be destroyed after six years.
The interviews included questions in the following domains:
Background on health system characteristics and structure
Chronic pain management and opioid prescribing guidelines implemented
Implementation strategies
Data systems available, including types of de-identified information
Efforts to address racial and ethnic disparities in chronic pain management, opioid prescribing, and access to MOUD
| File Type | application/vnd.openxmlformats-officedocument.wordprocessingml.document |
| Author | Angel, Karen C. (CDC/DDNID/NCIPC/OD) |
| File Modified | 0000-00-00 |
| File Created | 2022-02-18 |
© 2026 OMB.report | Privacy Policy